The Technology
MB-204
Marvel’s lead compound (MB-204) is a novel fluorinated derivative of Istradefylline, an FDA-approved adenosine A2a receptor antagonist. Designed to maximize therapeutic benefits, MB-204 is developed to work faster, more effectively, and with lower doses compared to existing treatments. Preclinical studies have demonstrated MB-204’s potential to address a range of conditions, including Alzheimer’s disease, depression, anxiety, and autism. MB-204 is currently being examined in preclinical models of Rett syndrome and Fragile X syndrome.

The compound’s mechanism of action involves modulating the A2A adenosine receptor, a well-validated target implicated in numerous neurological and psychiatric disorders. MB-204’s efficacy has been validated in independent laboratories, showing significant promise in reversing symptoms and underlying disease mechanisms. With completed cGMP synthesis, toxicology studies, and preparations for Phase 1 clinical trials, MB-204 is poised to become a game-changing therapy for patients suffering from these challenging conditions.
Emerging roles of dysregulated adenosine homeostasis in brain disorders with a specific focus on neurodegenerative diseases
Read full article here: https://pubmed.ncbi.nlm.nih.gov/34635103/
CD39-mediated ATP-adenosine signalling promotes hepatic stellate cell activation and alcoholic liver disease
Read full article here: https://pubmed.ncbi.nlm.nih.gov/34033815/
Proposed Development Timeline:
Q1 2026
MILESTONES
• cGMP drug product manufactured and released
• GLP toxicology studies completed in rats and dogs with acceptable safety margins (NOAEL)
• Non-dilutive funding secured for pediatric liquid formulation development (in progress; completion expected by end of Q1)
Q2 2026
NON-CLINICAL
Q4 2026
CLINICAL – Phase 1
NON-CLINICAL
Q1 2027
CLINICAL – Phase 1
• PK and exploratory biomarker analysis
NON-CLINICAL
*Note: timelines may be subject to change.
Marvel Non-Hallucinogenic Neuroplasticity Program
Marvel has identified a series of related compounds that appear to be potent, fast acting, water soluble and orally available small tryptamine derivatives that have anti-depressive activity but no overt hallucinatory activity in pilot studies.
Second Program: Safer Neuroplastic Promoting Drugs
Marvel has identified a set of molecules inspired by the tryptamine class of psychedelics that are:
- Fast acting anti-depressants
- Orally available
- Water soluble
- No evidence of hallucinations
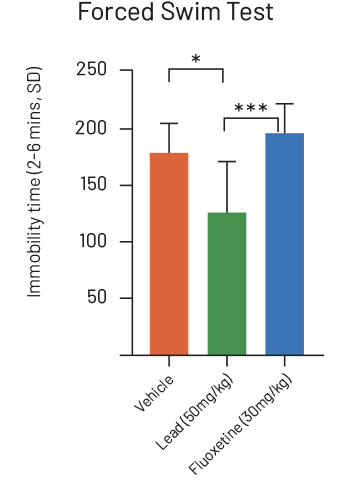
- Lead better than fluoxetine (Prozac, p<0.005)